
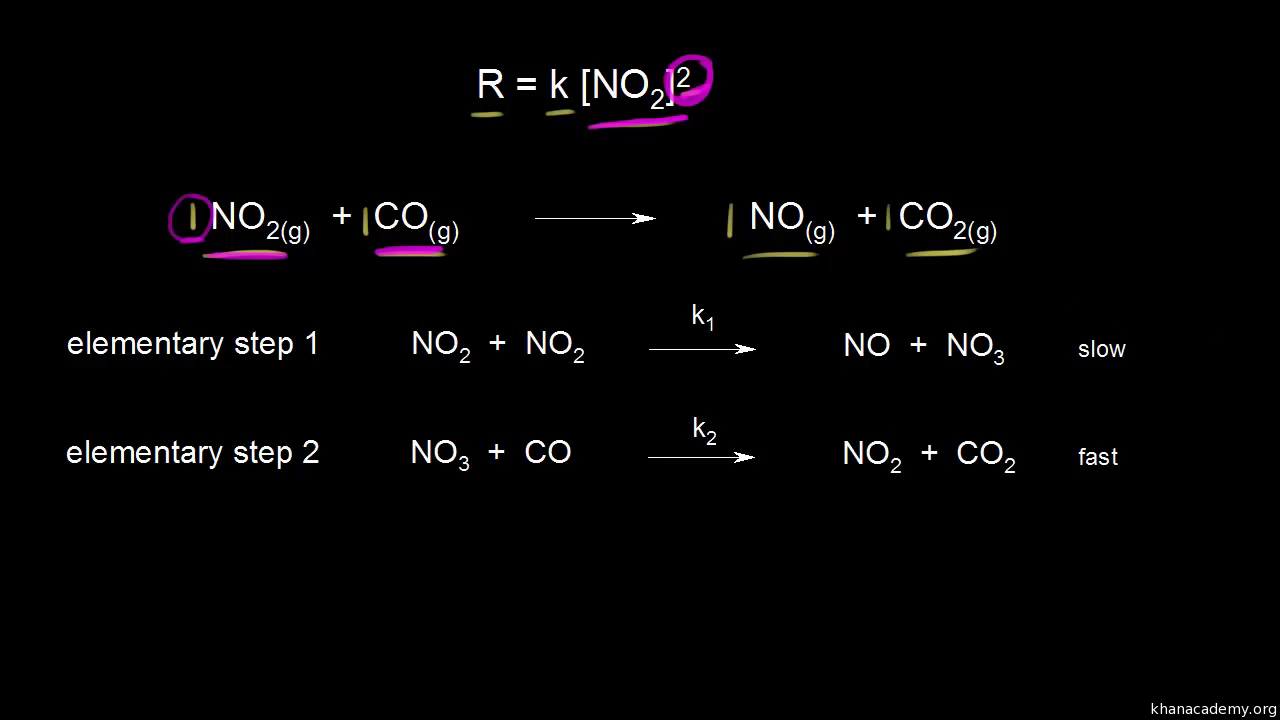
SOLVED: Consider the following mechanism step 1: 2A B+ C equilibrium step 2: B+ D 57 E slow overall: 2A+ D C+ E Determine the rate law for the overall reaction (where

Chapter 13: Chemical Kinetics CHE 124: General Chemistry II Dr. Jerome Williams, Ph.D. Saint Leo University. - ppt download

Writing Rate Laws of Reaction Mechanisms Using The Rate Determining Step - Chemical Kinetics - YouTube

How to Combine a Series of Elementary Reactions into an Overall Balanced Equation | Chemistry | Study.com

Determine the rate law for the overall reaction (where the overall rate constant is represented as k) - Home Work Help - Learn CBSE Forum

Finding the rate law from a rate-determining step that contains species not in the overall reaction : r/chemhelp
















![16.1/R2.2.6 Rate constant, overall order of reaction, order of reaction [HL IB Chemistry] - YouTube 16.1/R2.2.6 Rate constant, overall order of reaction, order of reaction [HL IB Chemistry] - YouTube](https://i.ytimg.com/vi/9sMFJMuZzmg/maxresdefault.jpg)